Voltage applied across a circuit causes electrons to move in one direction only. Electrons enter at the cathode, then flow out of the anode. Anode is positive and cathode is negative. Definition of Anode and CathodeĪnode and cathode are terms used in electric circuits. In special cases, electrodes need to be inert so they don’t react with solution ions or other species. Cathodes can be metallic ( zinc) or non-metallic ( carbon). For example, batteries powered by their own ‘alien charge’ with just one electrode.Īnodes are usually made from metals like copper or aluminum, as they conduct electricity well. 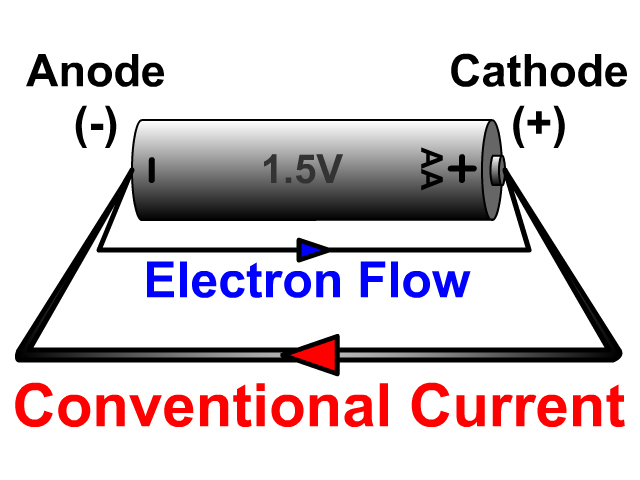
Although, some devices can be built using one type. Current flow rate, or amperage, depends on voltage and resistance across a circuit.Īn anode attracts positively charged particles and repels negative. An anode is where electrical current enters, and a cathode is where it exits. This creates a current that can be used to power devices.Anode and cathode are two electrode types used in electric circuits, and in processes such as electrolysis. When the battery is connected to a circuit, electrons flow from the cathode to the anode. The cathode is always connected to the negative terminal of the battery, and the anode is always connected to the positive terminal of the battery.The anode is usually made of a metal that forms a negative ion, such as copper or magnesium. The cathode is always made of a metal that forms a positive ion, such as zinc or aluminum. The cathode is the negative electrode and the anode is the positive electrode. The cell has two electrodes, called the cathode and the anode. A voltaic cell is a device that uses a chemical reaction to create an electrical current.The anode is at a higher potential than the cathode.Ĭharges of a Cathode and Anode of a Voltaic Cell The anode is the positively charged electrode. The cathode is negative and the anode is positive because the anode is the positive terminal of a battery and the cathode is the negative terminal.Reason Behind the Cathode as Negative and Anode as Positive In other words, the anode is the electrode where a substance loses electrons, while the cathode is the electrode where a substance gains electrons.The anode is the electrode at which oxidation occurs, while the cathode is the electrode at which reduction occurs.However, the table above highlights the general characteristics and distinctions between anode and cathode in most electrochemical systems. It’s important to note that the behavior of the anode and cathode can vary depending on the type of electrochemical cell or specific context. In a zinc-carbon battery, carbon acts as the cathode. In a zinc-carbon battery, zinc acts as the anode. In a galvanic cell, the cathode is connected to the positive terminal (cathode is the positive electrode).Īnode undergoes oxidation (loses electrons).Ĭathode undergoes reduction (gains electrons).Īnode may generate anions or release gas.Ĭathode may generate cations or consume gas. In a galvanic cell, the anode is connected to the negative terminal (anode is the negative electrode). In an electrolytic cell, the cathode is connected to the negative terminal (cathode is the negative electrode). In an electrolytic cell, the anode is connected to the positive terminal (anode is the positive electrode). The electrode where reduction (gain of electrons) occurs during an electrochemical reaction.Įlectrons flow from the anode to the cathode.Įlectrons flow towards the cathode from the anode. The electrode where oxidation (loss of electrons) occurs during an electrochemical reaction. Here’s a table highlighting the key differences between anode and cathode: In an electrochemical reaction, the anode is where oxidation occurs, meaning that electrons are lost from the anode and move to the cathode. An anode is an electrode in an electrochemical cell or battery that is positively charged and attracts negatively charged ions or electrons. In an electrolytic cell, it is the electrode at which electrons are transferred to the electrolyte. The anode is the electrode at which oxidation occurs. In an electrochemical reaction, the cathode is where reduction occurs, meaning that electrons are gained at the cathode. 
A cathode is an electrode in an electrochemical cell or battery that is negatively charged and attracts positively charged ions or electrons. Cathodes are usually negative with respect to an anode. What is Cathode?Ĭathode is the electrode through which current flows out of a device into the external circuit. Together, they form a galvanic cell or battery. In an electrochemical cell, an anode is the electrode at which oxidation occurs and a cathode is the electrode at which reduction occurs. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
